
Full resolution (TIFF) - On this page / på denna sida - Den thermochemiske Theori. I–II, af Julius Thomsen
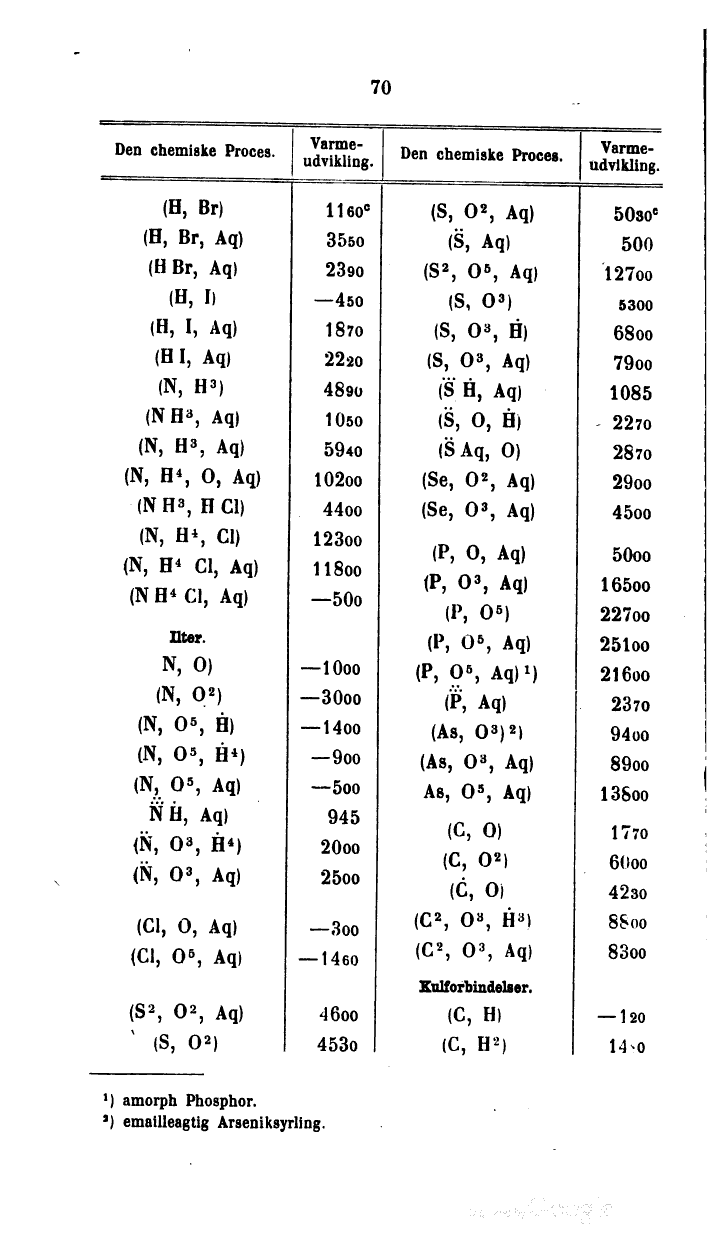
<< prev. page << föreg. sida << >> nästa sida >> next page >>
Below is the raw OCR text
from the above scanned image.
Do you see an error? Proofread the page now!
Här nedan syns maskintolkade texten från faksimilbilden ovan.
Ser du något fel? Korrekturläs sidan nu!
This page has been proofread at least once.
(diff)
(history)
Denna sida har korrekturlästs minst en gång.
(skillnad)
(historik)
| Den chemiske Proces. | Varmeudvikling. | Den chemiske Proces. | Varmeudvikling. |
| (H, Br) | 1160c | (S, O2, Aq) | 5030c |
| (H, Br, Aq) | 3550 | (S. ., Aq) | 500 |
| (H Br, Aq) | 2390 | (S2, O5, Aq) | 12700 |
| (H, I) | -450 | (S, O3) | 5300 |
| |H, I, Aq) | 1870 | (S, O3, H) | 6800 |
| (H I, Aq) | 2220 | (S, O3, Aq) | 7900 |
| (N, H3) | 4890 | (S. . . H., Aq) | 1085 |
| (N H3, Aq) | 1050 | (S. ., O, H) | 2270 |
| (N, H3, Aq) | 5940 | (S. . Aq, O) | 2870 |
| (N, H4, O, Aq) | 10200 | (Se, O2, Aq) | 2900 |
| (N H3, H Cl) | 4400 | (Se, O3, Aq) | 4500 |
| (N, H4, Cl) | 12300 | (P, O, Aq) | 5000 |
| (N, H4 Cl, Aq) | 1l800 | (P, O3, Aq) | 16500 |
| (N H4 Cl, Aq) | -500 | (P, O5) | 22700 |
| Ilter. | (P, O5, Aq) | 25100 | |
| N, 0) | -1000 | (P, O5, Aq)[1] | 2l600 |
| (N, O2) | -3000 | (P. . . . ., Aq) | 2370 |
| (N, O5, H) | -1400 | (As, O3)[2] | 9400 |
| (N, O5, H4) | -900 | (As, O3, Aq) | 8900 |
| (N, O5, Aq) | -500 | As, O5, Aq) | 13800 |
| N. . . . .H., Aq) | 945 | (C, O) | 1770 |
| (N. ., O3, H4) | 2000 | (C, O2) | 6000 |
| (N. ., O3, Aq) | 2500 | (C., O) | 4230 |
| (Cl, O, Aq) | -300 | (C2, O3, H.ⓛ3) | 8S00 |
| (Cl, O5, Aq) | -1460 | (C2, O3, Aq) | 8300 |
| (S2, O2, Aq) | 4600 | Kulforbindelser. | |
| (S, O2) | 4530 | (C, H) | -120 |
| (C, H2) | 1480 |
<< prev. page << föreg. sida << >> nästa sida >> next page >>